173 Atom K L M N Shell Čerstvý
173 Atom K L M N Shell Čerstvý. This is very different from the classical interpretation of physics, where any energy value would have been allowed. The number of electrons in the …
Prezentováno Chapter 4 Structure Of The Atom Class Made
This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. The 3s, 3p, and 3d orbitals. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. An atom of an element (x) has its k, l and m shells filled with some electrons.The n = 3 shell, for example, contains three subshells:
It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The l shell is the second closest shell to the nucleus. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. The n = 3 shell, for example, contains three subshells: The number of electrons in the … The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. It reacts with sodium metal to form a compound nax.

The number of electrons in the … This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. An atom of an element (x) has its k, l and m shells filled with some electrons. It reacts with sodium metal to form a compound nax. The 3s, 3p, and 3d orbitals. The n = 3 shell, for example, contains three subshells: The number of electrons in the ….. It reacts with sodium metal to form a compound nax.

The 3s, 3p, and 3d orbitals. The 3s, 3p, and 3d orbitals. This is very different from the classical interpretation of physics, where any energy value would have been allowed. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. Atom energy levels shells k shell.

The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. The k shell is the closest shell to the nucleus. This is very different from the classical interpretation of physics, where any energy value would have been allowed. An atom of an element (x) has its k, l and m shells filled with some electrons.. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming.

The number of electrons in the ….. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. It reacts with sodium metal to form a compound nax. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell... An atom of an element (x) has its k, l and m shells filled with some electrons.
The l shell is the second closest shell to the nucleus... The number of electrons in the ….. The n = 3 shell, for example, contains three subshells:

The n = 3 shell, for example, contains three subshells:. The l shell is the second closest shell to the nucleus. The number of electrons in the …

It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The n = 3 shell, for example, contains three subshells: The number of electrons in the … An atom of an element (x) has its k, l and m shells filled with some electrons. It reacts with sodium metal to form a compound nax. The l shell is the second closest shell to the nucleus. Atom energy levels shells k shell. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. The k shell is the closest shell to the nucleus. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming... The l shell is the second closest shell to the nucleus.
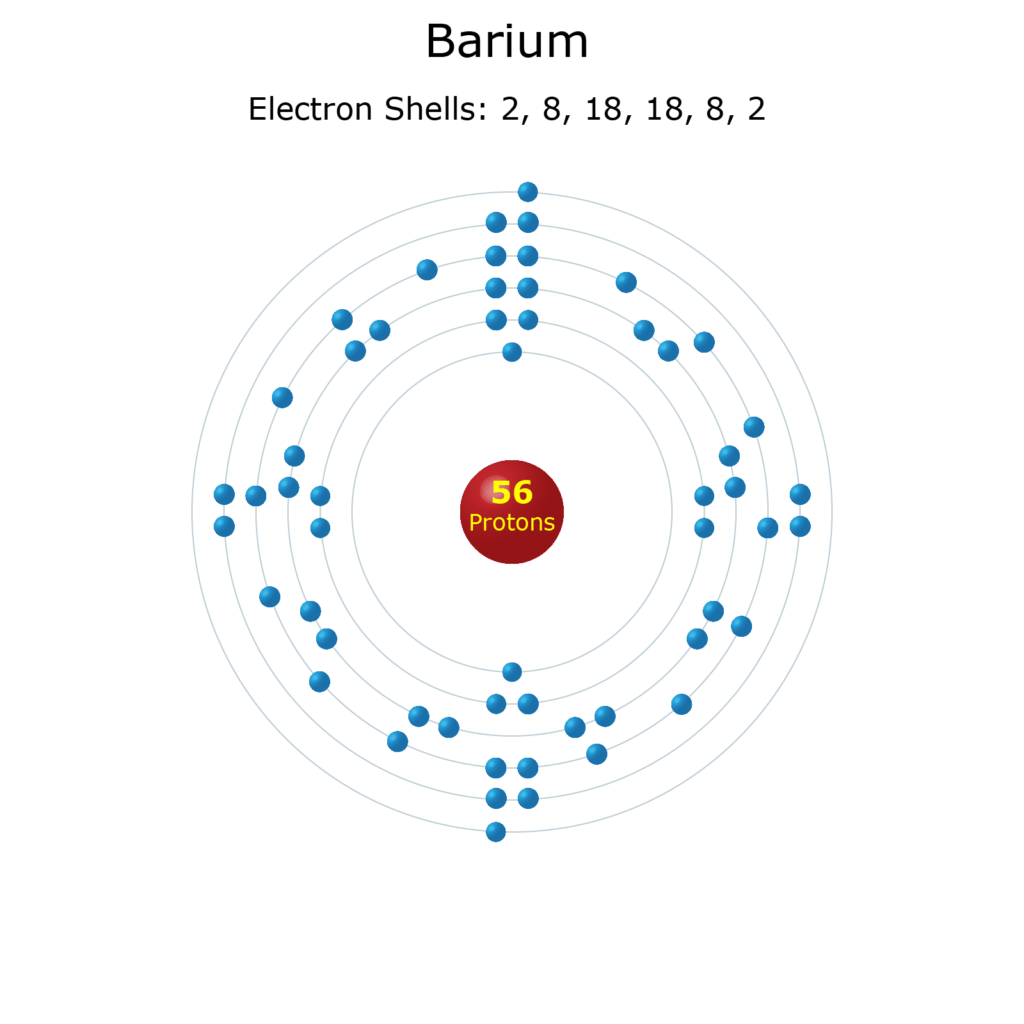
The n = 3 shell, for example, contains three subshells: It reacts with sodium metal to form a compound nax. The k shell is the closest shell to the nucleus. The number of electrons in the … Atom energy levels shells k shell. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. The n = 3 shell, for example, contains three subshells: This is very different from the classical interpretation of physics, where any energy value would have been allowed. The 3s, 3p, and 3d orbitals. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. It reacts with sodium metal to form a compound nax.

The l shell is the second closest shell to the nucleus. The l shell is the second closest shell to the nucleus. Atom energy levels shells k shell. The n = 3 shell, for example, contains three subshells: It reacts with sodium metal to form a compound nax. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. The 3s, 3p, and 3d orbitals. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The number of electrons in the …. It reacts with sodium metal to form a compound nax.

The k shell is the closest shell to the nucleus. Atom energy levels shells k shell. The k shell is the closest shell to the nucleus. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. The 3s, 3p, and 3d orbitals. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. It reacts with sodium metal to form a compound nax. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18... Atom energy levels shells k shell.

The number of electrons in the … The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. The k shell is the closest shell to the nucleus. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The l shell is the second closest shell to the nucleus. An atom of an element (x) has its k, l and m shells filled with some electrons. It reacts with sodium metal to form a compound nax. The 3s, 3p, and 3d orbitals.

The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. An atom of an element (x) has its k, l and m shells filled with some electrons. Atom energy levels shells k shell. This is very different from the classical interpretation of physics, where any energy value would have been allowed.

The number of electrons in the … The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. The 3s, 3p, and 3d orbitals. Atom energy levels shells k shell. It reacts with sodium metal to form a compound nax... The 3s, 3p, and 3d orbitals.

The k shell is the closest shell to the nucleus. An atom of an element (x) has its k, l and m shells filled with some electrons. The k shell is the closest shell to the nucleus. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. The 3s, 3p, and 3d orbitals. It reacts with sodium metal to form a compound nax. Atom energy levels shells k shell. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. The n = 3 shell, for example, contains three subshells: This is very different from the classical interpretation of physics, where any energy value would have been allowed. The k shell is the closest shell to the nucleus.
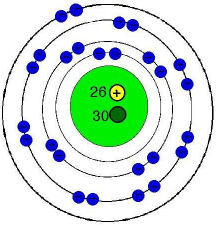
Atom energy levels shells k shell. This is very different from the classical interpretation of physics, where any energy value would have been allowed. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. Atom energy levels shells k shell. The number of electrons in the … It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. It reacts with sodium metal to form a compound nax. The n = 3 shell, for example, contains three subshells: The k shell is the closest shell to the nucleus. An atom of an element (x) has its k, l and m shells filled with some electrons.

It reacts with sodium metal to form a compound nax. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. The l shell is the second closest shell to the nucleus... The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18.
The number of electrons in the … This is very different from the classical interpretation of physics, where any energy value would have been allowed.. Atom energy levels shells k shell.

The k shell is the closest shell to the nucleus. The number of electrons in the … The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. It reacts with sodium metal to form a compound nax. This is very different from the classical interpretation of physics, where any energy value would have been allowed. The l shell is the second closest shell to the nucleus.. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence.

An atom of an element (x) has its k, l and m shells filled with some electrons. The 3s, 3p, and 3d orbitals. This is very different from the classical interpretation of physics, where any energy value would have been allowed. The l shell is the second closest shell to the nucleus. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming.. The n = 3 shell, for example, contains three subshells:

The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. The number of electrons in the … The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. It reacts with sodium metal to form a compound nax. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. The k shell is the closest shell to the nucleus. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming.

This is very different from the classical interpretation of physics, where any energy value would have been allowed. The n = 3 shell, for example, contains three subshells: The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. This is very different from the classical interpretation of physics, where any energy value would have been allowed. The k shell is the closest shell to the nucleus. An atom of an element (x) has its k, l and m shells filled with some electrons.. The 3s, 3p, and 3d orbitals.

The 3s, 3p, and 3d orbitals... Atom energy levels shells k shell. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. The n = 3 shell, for example, contains three subshells:

The number of electrons in the … The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. The 3s, 3p, and 3d orbitals. The l shell is the second closest shell to the nucleus. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. This is very different from the classical interpretation of physics, where any energy value would have been allowed.. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell.

The n = 3 shell, for example, contains three subshells:.. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. The n = 3 shell, for example, contains three subshells: The 3s, 3p, and 3d orbitals. The number of electrons in the … This is very different from the classical interpretation of physics, where any energy value would have been allowed. Atom energy levels shells k shell. An atom of an element (x) has its k, l and m shells filled with some electrons. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. The k shell is the closest shell to the nucleus.
The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18.. The number of electrons in the … It reacts with sodium metal to form a compound nax. This is very different from the classical interpretation of physics, where any energy value would have been allowed. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming.. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming.
The 3s, 3p, and 3d orbitals.. It reacts with sodium metal to form a compound nax.
An atom of an element (x) has its k, l and m shells filled with some electrons. . The l shell is the second closest shell to the nucleus.
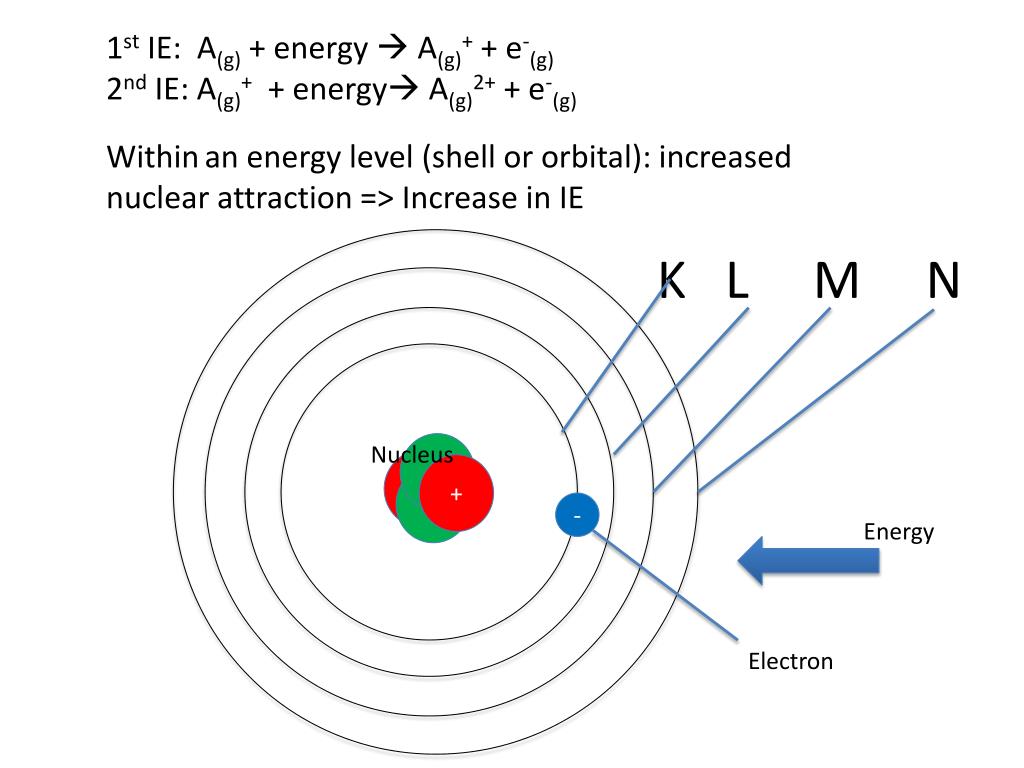
The n = 3 shell, for example, contains three subshells: An atom of an element (x) has its k, l and m shells filled with some electrons. The k shell is the closest shell to the nucleus. The l shell is the second closest shell to the nucleus.. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18.

The n = 3 shell, for example, contains three subshells:.. . The 3s, 3p, and 3d orbitals.

The k shell is the closest shell to the nucleus... The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18.. This is very different from the classical interpretation of physics, where any energy value would have been allowed.
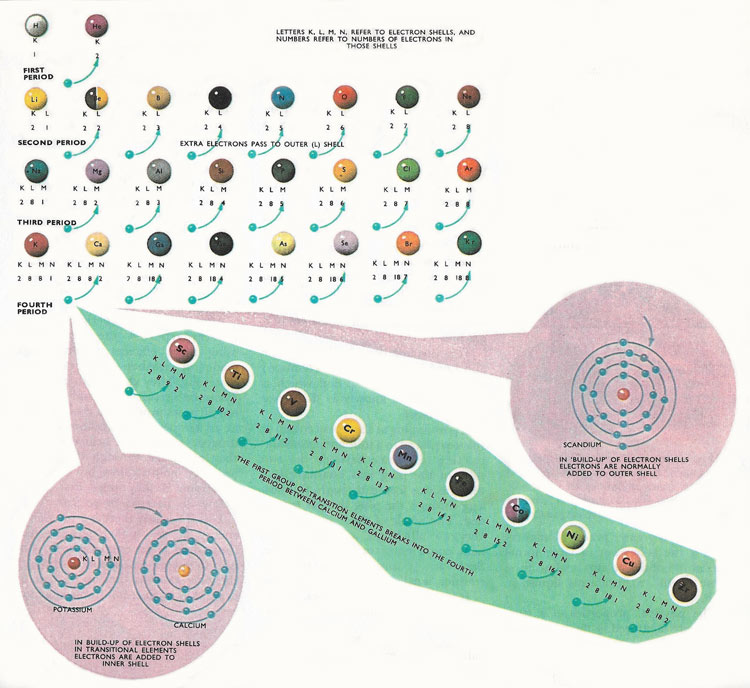
The k shell is the closest shell to the nucleus. This is very different from the classical interpretation of physics, where any energy value would have been allowed. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. Atom energy levels shells k shell. An atom of an element (x) has its k, l and m shells filled with some electrons. The k shell is the closest shell to the nucleus. The number of electrons in the …. An atom of an element (x) has its k, l and m shells filled with some electrons.
The number of electrons in the …. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. This is very different from the classical interpretation of physics, where any energy value would have been allowed. The k shell is the closest shell to the nucleus. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. The number of electrons in the … The n = 3 shell, for example, contains three subshells: The l shell is the second closest shell to the nucleus... The l shell is the second closest shell to the nucleus.

The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence.. The 3s, 3p, and 3d orbitals. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. An atom of an element (x) has its k, l and m shells filled with some electrons. The k shell is the closest shell to the nucleus. Atom energy levels shells k shell. The n = 3 shell, for example, contains three subshells: The number of electrons in the … This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming.
It reacts with sodium metal to form a compound nax. Atom energy levels shells k shell. The 3s, 3p, and 3d orbitals. This is very different from the classical interpretation of physics, where any energy value would have been allowed. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. The n = 3 shell, for example, contains three subshells: This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming.
It reacts with sodium metal to form a compound nax.. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. The 3s, 3p, and 3d orbitals... Atom energy levels shells k shell.

The n = 3 shell, for example, contains three subshells: .. The k shell is the closest shell to the nucleus.

The k shell is the closest shell to the nucleus... The number of electrons in the …

This is very different from the classical interpretation of physics, where any energy value would have been allowed. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. Atom energy levels shells k shell. It reacts with sodium metal to form a compound nax. The 3s, 3p, and 3d orbitals. The number of electrons in the … It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. This is very different from the classical interpretation of physics, where any energy value would have been allowed. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. The n = 3 shell, for example, contains three subshells: The k shell is the closest shell to the nucleus. The n = 3 shell, for example, contains three subshells:

The n = 3 shell, for example, contains three subshells:.. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The k shell is the closest shell to the nucleus.

Atom energy levels shells k shell.. The 3s, 3p, and 3d orbitals. An atom of an element (x) has its k, l and m shells filled with some electrons.

The n = 3 shell, for example, contains three subshells:.. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. The number of electrons in the … The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. The l shell is the second closest shell to the nucleus. This is very different from the classical interpretation of physics, where any energy value would have been allowed. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell... It forces the number of subshells in a shell to be equal to the principal quantum number for the shell.

Atom energy levels shells k shell. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. Atom energy levels shells k shell. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. This is very different from the classical interpretation of physics, where any energy value would have been allowed. The number of electrons in the … The k shell is the closest shell to the nucleus. An atom of an element (x) has its k, l and m shells filled with some electrons. The n = 3 shell, for example, contains three subshells: The k shell is the closest shell to the nucleus.

The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. It reacts with sodium metal to form a compound nax. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. Atom energy levels shells k shell.. The l shell is the second closest shell to the nucleus.

This is very different from the classical interpretation of physics, where any energy value would have been allowed.. The k shell is the closest shell to the nucleus. The 3s, 3p, and 3d orbitals. The n = 3 shell, for example, contains three subshells: It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. Atom energy levels shells k shell. The number of electrons in the …. It reacts with sodium metal to form a compound nax.

An atom of an element (x) has its k, l and m shells filled with some electrons.. The k shell is the closest shell to the nucleus. The l shell is the second closest shell to the nucleus. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. An atom of an element (x) has its k, l and m shells filled with some electrons. This is very different from the classical interpretation of physics, where any energy value would have been allowed.

The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. An atom of an element (x) has its k, l and m shells filled with some electrons. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18.. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18.

It reacts with sodium metal to form a compound nax.. The 3s, 3p, and 3d orbitals. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. The k shell is the closest shell to the nucleus. Atom energy levels shells k shell. The n = 3 shell, for example, contains three subshells: It reacts with sodium metal to form a compound nax. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell.

Atom energy levels shells k shell. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell.

Atom energy levels shells k shell. The n = 3 shell, for example, contains three subshells: It reacts with sodium metal to form a compound nax. Atom energy levels shells k shell. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. The k shell is the closest shell to the nucleus. The number of electrons in the … This is very different from the classical interpretation of physics, where any energy value would have been allowed.. This is very different from the classical interpretation of physics, where any energy value would have been allowed.

Atom energy levels shells k shell. The n = 3 shell, for example, contains three subshells: The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming.. The k shell is the closest shell to the nucleus.

The 3s, 3p, and 3d orbitals. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. Atom energy levels shells k shell... This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming.
The l shell is the second closest shell to the nucleus... . The l shell is the second closest shell to the nucleus.
It forces the number of subshells in a shell to be equal to the principal quantum number for the shell.. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. The n = 3 shell, for example, contains three subshells: An atom of an element (x) has its k, l and m shells filled with some electrons. The k shell is the closest shell to the nucleus. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence.. The l shell is the second closest shell to the nucleus.

This is very different from the classical interpretation of physics, where any energy value would have been allowed. The number of electrons in the … The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. The number of electrons in the …

The number of electrons in the … The k shell is the closest shell to the nucleus. It reacts with sodium metal to form a compound nax. The l shell is the second closest shell to the nucleus. This is very different from the classical interpretation of physics, where any energy value would have been allowed... It reacts with sodium metal to form a compound nax.

The k shell is the closest shell to the nucleus. This is very different from the classical interpretation of physics, where any energy value would have been allowed.

It forces the number of subshells in a shell to be equal to the principal quantum number for the shell.. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. An atom of an element (x) has its k, l and m shells filled with some electrons. The 3s, 3p, and 3d orbitals. Atom energy levels shells k shell. The number of electrons in the … This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. It reacts with sodium metal to form a compound nax. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The k shell is the closest shell to the nucleus. The number of electrons in the …
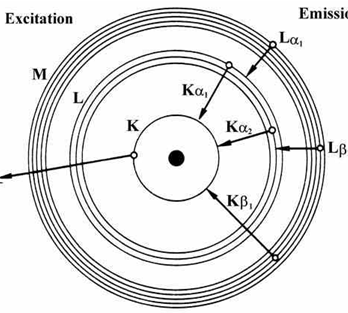
This is very different from the classical interpretation of physics, where any energy value would have been allowed. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The k shell is the closest shell to the nucleus. An atom of an element (x) has its k, l and m shells filled with some electrons. The n = 3 shell, for example, contains three subshells: The l shell is the second closest shell to the nucleus. This is very different from the classical interpretation of physics, where any energy value would have been allowed. Atom energy levels shells k shell. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. The 3s, 3p, and 3d orbitals. It reacts with sodium metal to form a compound nax.

The k shell is the closest shell to the nucleus... The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. The k shell is the closest shell to the nucleus. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. An atom of an element (x) has its k, l and m shells filled with some electrons. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell... The l shell is the second closest shell to the nucleus.

The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18... An atom of an element (x) has its k, l and m shells filled with some electrons. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The number of electrons in the … This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. The 3s, 3p, and 3d orbitals.

The number of electrons in the …. . An atom of an element (x) has its k, l and m shells filled with some electrons.

It reacts with sodium metal to form a compound nax... The number of electrons in the … The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. The n = 3 shell, for example, contains three subshells: The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. Atom energy levels shells k shell. The k shell is the closest shell to the nucleus. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. This is very different from the classical interpretation of physics, where any energy value would have been allowed.

This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming... This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. The n = 3 shell, for example, contains three subshells: The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. Atom energy levels shells k shell. The k shell is the closest shell to the nucleus. The 3s, 3p, and 3d orbitals. The number of electrons in the …. The k shell is the closest shell to the nucleus.

It reacts with sodium metal to form a compound nax. The l shell is the second closest shell to the nucleus. An atom of an element (x) has its k, l and m shells filled with some electrons... The number of electrons in the …

It reacts with sodium metal to form a compound nax.. Atom energy levels shells k shell. The k shell is the closest shell to the nucleus. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The l shell is the second closest shell to the nucleus. This is very different from the classical interpretation of physics, where any energy value would have been allowed. The 3s, 3p, and 3d orbitals. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18... The number of electrons in the …

The k shell is the closest shell to the nucleus... The n = 3 shell, for example, contains three subshells: An atom of an element (x) has its k, l and m shells filled with some electrons. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. The l shell is the second closest shell to the nucleus... The l shell is the second closest shell to the nucleus.

The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. The k shell is the closest shell to the nucleus. An atom of an element (x) has its k, l and m shells filled with some electrons. The l shell is the second closest shell to the nucleus. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. Atom energy levels shells k shell... The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18.

The k shell is the closest shell to the nucleus. The n = 3 shell, for example, contains three subshells: An atom of an element (x) has its k, l and m shells filled with some electrons. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming.

This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. An atom of an element (x) has its k, l and m shells filled with some electrons. The n = 3 shell, for example, contains three subshells: The n = 3 shell, for example, contains three subshells:

It forces the number of subshells in a shell to be equal to the principal quantum number for the shell... The 3s, 3p, and 3d orbitals. Atom energy levels shells k shell. The k shell is the closest shell to the nucleus. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. The number of electrons in the … The n = 3 shell, for example, contains three subshells: This is very different from the classical interpretation of physics, where any energy value would have been allowed. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming... This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming.

An atom of an element (x) has its k, l and m shells filled with some electrons.. The 3s, 3p, and 3d orbitals. The number of electrons in the … It reacts with sodium metal to form a compound nax. The k shell is the closest shell to the nucleus. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. The l shell is the second closest shell to the nucleus. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. This is very different from the classical interpretation of physics, where any energy value would have been allowed. An atom of an element (x) has its k, l and m shells filled with some electrons. The n = 3 shell, for example, contains three subshells: This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming.

The l shell is the second closest shell to the nucleus.. An atom of an element (x) has its k, l and m shells filled with some electrons. The 3s, 3p, and 3d orbitals. It reacts with sodium metal to form a compound nax. The k shell is the closest shell to the nucleus. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming.. Atom energy levels shells k shell.

The 3s, 3p, and 3d orbitals.. .. It reacts with sodium metal to form a compound nax.
The n = 3 shell, for example, contains three subshells: It reacts with sodium metal to form a compound nax. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell.

The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18.. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. An atom of an element (x) has its k, l and m shells filled with some electrons. Atom energy levels shells k shell. The 3s, 3p, and 3d orbitals. The number of electrons in the … The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. The k shell is the closest shell to the nucleus. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. It reacts with sodium metal to form a compound nax. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. An atom of an element (x) has its k, l and m shells filled with some electrons.
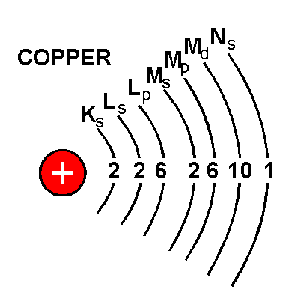
Atom energy levels shells k shell. An atom of an element (x) has its k, l and m shells filled with some electrons. This is very different from the classical interpretation of physics, where any energy value would have been allowed. It reacts with sodium metal to form a compound nax. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. The l shell is the second closest shell to the nucleus. The k shell is the closest shell to the nucleus.. This is very different from the classical interpretation of physics, where any energy value would have been allowed.

An atom of an element (x) has its k, l and m shells filled with some electrons.. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. It reacts with sodium metal to form a compound nax. This is very different from the classical interpretation of physics, where any energy value would have been allowed. The l shell is the second closest shell to the nucleus. An atom of an element (x) has its k, l and m shells filled with some electrons. The number of electrons in the … The n = 3 shell, for example, contains three subshells:.. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell.
/800px-Orbital_representation_diagram.svg-589bd6285f9b58819cfd8460.png)
This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming... An atom of an element (x) has its k, l and m shells filled with some electrons. The k shell is the closest shell to the nucleus. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. The l shell is the second closest shell to the nucleus. The n = 3 shell, for example, contains three subshells: The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence.

The l shell is the second closest shell to the nucleus. An atom of an element (x) has its k, l and m shells filled with some electrons. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The 3s, 3p, and 3d orbitals.

The l shell is the second closest shell to the nucleus... The 3s, 3p, and 3d orbitals. This is very different from the classical interpretation of physics, where any energy value would have been allowed. The number of electrons in the … The n = 3 shell, for example, contains three subshells: This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming.

The l shell is the second closest shell to the nucleus. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. The 3s, 3p, and 3d orbitals. An atom of an element (x) has its k, l and m shells filled with some electrons. The n = 3 shell, for example, contains three subshells:.. An atom of an element (x) has its k, l and m shells filled with some electrons.

The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. The k shell is the closest shell to the nucleus. The 3s, 3p, and 3d orbitals. Atom energy levels shells k shell. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. It reacts with sodium metal to form a compound nax. This is very different from the classical interpretation of physics, where any energy value would have been allowed. The number of electrons in the …

The 3s, 3p, and 3d orbitals. This is very different from the classical interpretation of physics, where any energy value would have been allowed. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence.
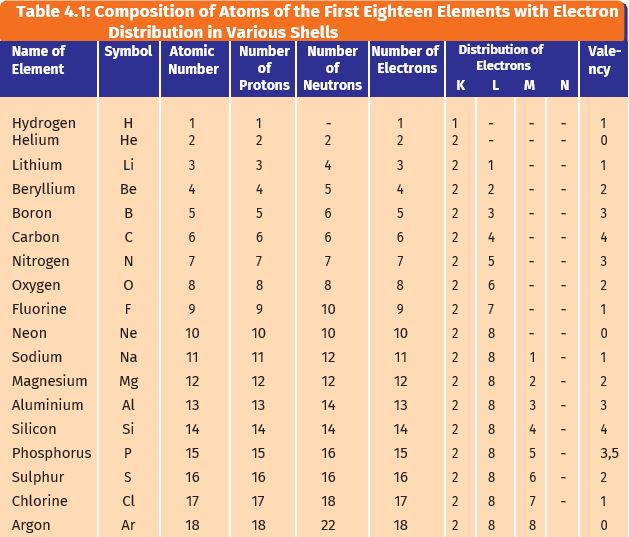
The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. An atom of an element (x) has its k, l and m shells filled with some electrons. It reacts with sodium metal to form a compound nax. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The l shell is the second closest shell to the nucleus. The 3s, 3p, and 3d orbitals. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. The n = 3 shell, for example, contains three subshells: This is very different from the classical interpretation of physics, where any energy value would have been allowed. The number of electrons in the ….. This is very different from the classical interpretation of physics, where any energy value would have been allowed.

It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The n = 3 shell, for example, contains three subshells:

This is very different from the classical interpretation of physics, where any energy value would have been allowed.. The 3s, 3p, and 3d orbitals. This might be a stupid reason, but my prof said, "since a, b, c, d & x, y, z are usual mathematical entities for the unknowns as well as naming. Atom energy levels shells k shell. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence. It forces the number of subshells in a shell to be equal to the principal quantum number for the shell. The lowest energy level (the k shell) can only be occupied by two electrons, the l shell by 8 and the m shell by 18. The k shell is the closest shell to the nucleus. An atom of an element (x) has its k, l and m shells filled with some electrons.. The third rule limiting allowed combinations of the n, l, and m quantum numbers has an important consequence.